
The Next Generation OCT Skin Imaging

15-second scan • >1 mm imaging depth • 6 × 6 mm field of view • AI-assisted interpretation
Next-generation
OCT skin imaging
High resolution, non-invasive imaging of skin structure & function- in only seconds.
VivoSight Dx Pro delivers unparalleled, in-vivo visualisation of the skin, enabling confident clinical insight and accelerated research without the need for biopsy.
- Unrivalled >1mm depth penetration giving high-resolution, 3D, skin imaging
- Widest field-of-view, 6mm x 6mm scan enables visualization of entire lesion
- Completely non-invasive. Better accepted by your patients/subjects
- Fast, efficient scanning. Full 3D imaging in just 15 seconds
Highly cost-effective – significantly reduces time, and costs across both clinical workflows and research applications.
How it Works
An optical analogue to ultrasound imaging using lasers
Optical Coherence Tomography (OCT) is an imaging technology based on light rather than sound. In much the same way that ultrasound uses acoustic waves, VivoSight OCT employs eye-safe, infrared scanning laser light to generate a high-resolution, three-dimensional block of image data.
With its unrivalled 6 mm × 6 mm field of view and >1 mm imaging depth, VivoSight Dx Pro allows clinicians and researchers to visualise deep into the dermis, capturing the full subsurface architecture of the lesion. Powerful, intuitive software transforms complex OCT data into clear, easy-to-interpret 3D images.

VivoSight’s >1mm depth and 6x6mm field-of-view, compared with other OCT system
Dynamic OCT (D-OCT)
Functional insight beyond structure.
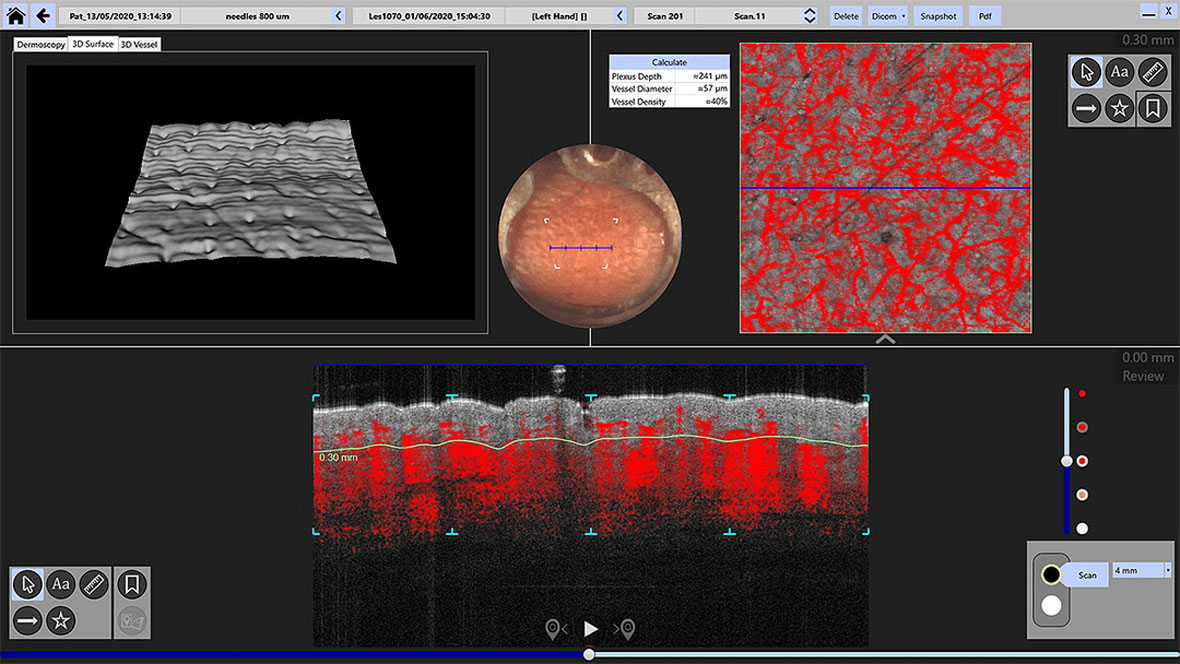
Dynamic OCT (D-OCT) extends conventional structural OCT by visualising real- time microvascular blood flow within the skin, adding functional information to high-resolution morphology. By detecting motion contrast from flowing blood cells, D-OCT reveals vascular patterns and perfusion changes associated with inflammation, tumour activity, and treatment response — without contrast agents or invasive procedures.
Fully integrated into the VivoSight Dx Pro workflow, D-OCT supports more confident lesion characterisation, aids differentiation of benign and malignant features, and enables objective monitoring of therapeutic effect over time, all from the same fast, non-invasive scan.
3-D rendered views optimize visualization of skin micro-vasculature
Harness the Power of AI
Making OCT accessible to every dermatologist.
VivoSight Dx Pro is designed to be easier to learn and faster to use than ever before. With VivoAID*, AI-driven image markers are automatically placed to support interpretation, consistency, and training — helping users build confidence quickly.
Learn more about VivoAID and its role in NMSC diagnosis
VivoAID AI-assistance speeds and eases the OCT-interpretation learning curve
Comprehensive,
quantitative skin analysis
Objective metrics at the click of a button
VivoTools* provides rapid, automated calculation of key quantitative skin parameters, enabling objective skin health assessment and comparison over time. From clinical research to product development, VivoTools turns OCT images into actionable data.
Discover how VivoTools can accelerate your skin research or aesthetic product development
*VivoTools is for investigational use only. Not for use in the clinical pathway.
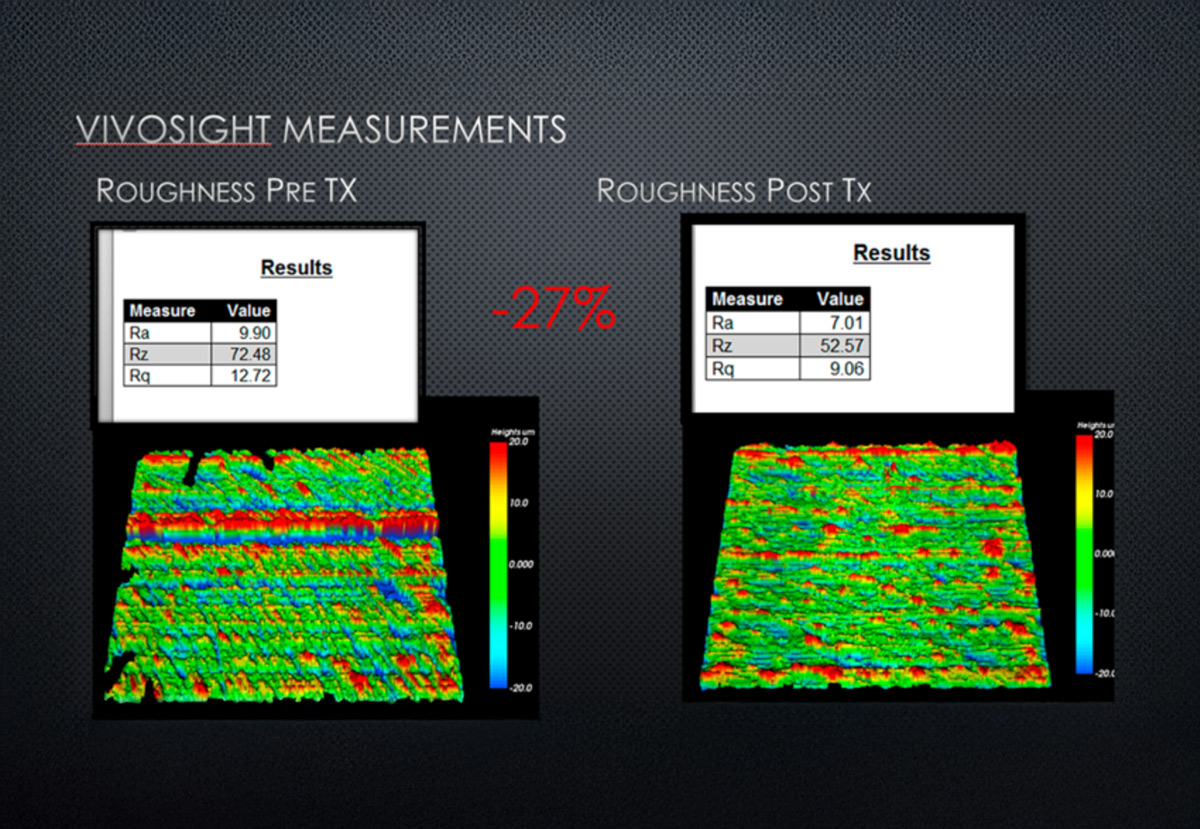
VivoTools pre- and post-treatment skin roughness quantification
*VivoAID and VivoTools are available as extra cost software options

Interested? Schedule a Demo Today!
Our team is available for your online demo of VivoSight skin imaging. Complete the form in the link below to schedule a time with one of our experts.

Explore Further
-
Clinical Applications
Learn more about VivoSight clinical applications
-
Research Applications
Learn more about VivoSight research applications
VivoSight Dx Pro is CE-marked as a Class IIa Medical Device under MDR (EU 2017/745)
VivoSight Dx is intended for imaging of the skin and nails of the human body, to provide cross-sectional images of the tissue structure and blood vessel network.
VivoSight Dx is indicated for use:
- As an adjunct device to support the clinical non-invasive diagnosis and follow-up of low-risk superficial BCC and nodular BCC, for clinically unclear nonpigmented lesions < 1 cm diameter in low-risk anatomical areas.
- As an adjunct device to support the clinical non-invasive differential diagnosis of Actinic Keratosis versus Bowen’s Disease, Squamous Cell Carcinoma and Basal Cell Carcinoma, for clinically unclear nonpigmented lesions suspected for Actinic Keratosis
VivoAID
VivoAID is provided solely as an educational aid. OCT image analysis and interpretation remain the responsibility of the physician.

